
Why Eyedrop Filling Lines Fail Leak Tests: The Hidden Role of Torque Control
In many pharmaceutical production lines, filling accuracy is often the main focus when selecting an eyedrop filling system. However, one critical factor is frequently underestimated — torque control during the capping process.
Improper torque settings can lead to several serious issues:
- Leaking bottles
- Cap damage
- Product contamination risks
- Failed transportation tests
- Inconsistent product quality
For sterile ophthalmic products, even small deviations in torque can affect container integrity and product sterility.
Understanding how torque control works is therefore essential when evaluating an eyedrop filling line.
Two Common Torque Control Methods in Eyedrop Filling Systems
Modern capping systems generally use two main torque control approaches:
- Mechanical clutch torque control
- Servo torque control
Both methods are widely used in pharmaceutical packaging equipment, and each has advantages depending on the application.
Mechanical Clutch Torque Control
Many eyedrop filling lines still rely on mechanical clutch torque control.
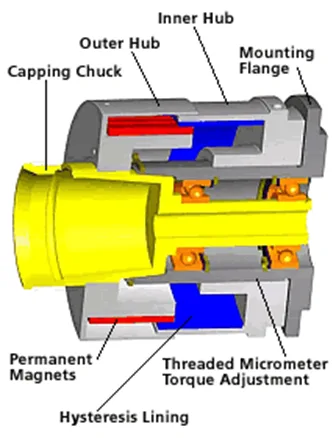
In this system, torque is controlled using mechanical springs and clutch components inside the capping head.
Mechanical clutch systems are commonly used because they are:
- Simple in design
- Cost-effective
- Widely understood by operators
However, they also have limitations.
Over time:
- Springs can wear out
- Torque settings may gradually drift
- Operators must manually adjust torque
Another important limitation is that torque data is typically not recorded.
For pharmaceutical environments operating under GMP requirements, the lack of torque traceability can become a concern.
If a leakage issue occurs later, it may be difficult to verify what torque was applied during production.
Servo Torque Control in Modern Capping Systems
Some modern ophthalmic filling lines use servo-driven torque control instead of mechanical clutch systems.
Servo-controlled capping heads regulate torque using motor current and programmable parameters.
This allows torque settings to be controlled digitally rather than mechanically.
Servo torque control offers several advantages:
- Highly repeatable torque adjustment
- Real-time torque monitoring for each bottle
- Programmable torque recipes for different containers
- Automatic detection of under-torque conditions
In addition, torque values can be recorded as part of the Electronic Batch Record (EBR).
This traceability can be particularly valuable in regulated pharmaceutical environments.
When Servo Torque Control Is Especially Useful
Servo-controlled torque systems can be particularly beneficial when:
- Multiple bottle formats are used
- Frequent product changeovers occur
- Torque documentation is required for GMP compliance
- High consistency is required for sterile products
Instead of manually adjusting mechanical components, operators can simply select a predefined torque recipe.
This improves consistency and reduces operator error.
Learn more about our aseptic ophthalmic filling system.
When Mechanical Clutch Systems Still Make Sense
Although servo torque control offers advanced monitoring and documentation, mechanical clutch systems still have their place.
They may remain suitable when:
- Project budgets are limited
- Production requirements are relatively simple
- Torque traceability is not required
Both technologies are widely used in pharmaceutical packaging.
The key is choosing the most appropriate torque control method for the application.
Conclusion
In an eyedrop filling line, filling accuracy alone does not guarantee reliable product quality. Proper torque control during the capping process plays an equally important role in maintaining container integrity and preventing leakage.
Mechanical clutch systems remain suitable for certain applications, while servo-controlled systems provide greater precision, monitoring capability, and documentation for GMP environments.
Manufacturers evaluating an ophthalmic filling line should therefore consider torque control methods carefully to ensure consistent performance and regulatory compliance.
Looking for a reliable ophthalmic filling solution?
MH Multipack designs advanced pharmaceutical filling and capping systems for sterile ophthalmic products. Our team can help you evaluate torque control options and select the right solution for your production requirements.
Frequently Asked Questions (FAQs)
Why do eyedrop bottles sometimes leak after the filling and capping process?
Leakage often occurs when the capping torque is not properly controlled. If the applied torque is too low, the cap may not seal tightly. If the torque is too high, the cap or bottle thread may deform, affecting seal integrity and causing leakage during transport or storage.
What is the ideal torque for eyedrop bottle caps?
The ideal torque depends on the bottle material, cap design, thread type, and product requirements. Pharmaceutical manufacturers typically validate torque ranges during packaging qualification to ensure proper sealing, product sterility, and patient usability.
What is the difference between mechanical clutch and servo torque control?
Mechanical clutch systems regulate torque using springs and mechanical components, while servo torque control uses motors and programmable parameters. Servo systems offer higher repeatability, real-time torque monitoring, and digital recording of torque data for GMP environments.
Why is torque monitoring important in pharmaceutical production?
Torque monitoring helps ensure consistent cap sealing and provides traceable data for quality assurance. In regulated environments, recorded torque data can support GMP documentation, validation, and investigation if packaging issues occur.
When should manufacturers consider servo torque control for capping systems?
Servo torque control is particularly useful when production requires high consistency, multiple container formats, frequent changeovers, or electronic batch records for regulatory compliance.
Disclaimer:
The information provided in this article is for general informational purposes only. Readers are encouraged to consult with a qualified industry professional or contact MH Multipack for advice specific to their operations, regulatory compliance, or equipment needs. While we strive to provide accurate and up-to-date information, we cannot guarantee they are entirely error-free. MH Multipack Sdn Bhd assumes no responsibility for any errors or omissions.


